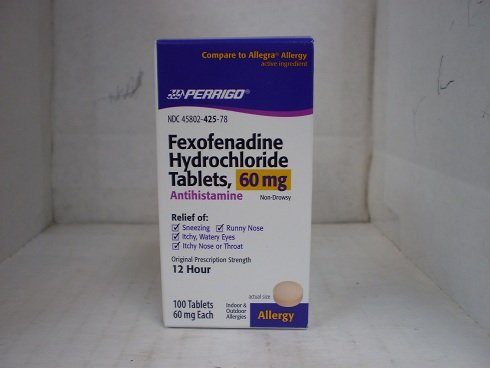
Fexofenadine 60 mg Tablets Generic Allegra - 100 Count Bottle. Toggle 1. Sign In; Sign Up; Help. 800-748-7001; 888-870-2808; Help Center; Fexofenadine HCl.
Adults and children over age 12 with allergies may take 60 mg or 180 mg fexofenadine tablets. This eMedTV article provides fexofenadine dosing guidelines for adults.

Allegra12 Hour Allergy, 60mg Tablets at Walgreens. Get free shipping at 35 and view promotions and reviews for Allegra12 Hour Allergy, 60mg Tablets.
Adults and children 12 years old and older being treated for allergies may take either the 60 mg tablets (one tablet, twice a day) or the 180 mg tablets (one tablet, once daily). The recommended fexofenadine dosage for children 6 to 11 years old is 30 mg twice a day (which can be taken as orally disintegrating tablets or oral suspension). For children two to five years old, the recommended dose is 30 mg (5 mL or one teaspoonful) of the oral suspension twice daily.
Buy Walgreens Fexofenadine Hcl 60Mg Tablets with free shipping on orders over 35, low prices product reviews drugstore.com.
Fexofenadine official prescribing information for healthcare professionals. Includes: indications, dosage, adverse reactions, pharmacology and more.
For seasonal allergies, adults or children over age 12 usually take Allegra 60 mg tablets or 180 mg tablets.
Seasonal Allergic Rhinitis and Chronic Idiopathic Urticaria Adults and Children 12 Years and Older: The recommended dose of Fexofenadine hydrochloride tablets USP are 60 mg twice daily or 180 mg once daily with water. A dose of 60 mg once daily is recommended as the starting dose in patients with decreased renal function [see Clinical Pharmacology (12.3)]. Slideshow: What's That? 10 Of The Most Common Skin Conditions Children 6 to 11 Years: The recommended dose of Fexofenadine hydrochloride tablets USP are 30 mg twice daily with water. A dose of 30 mg once daily is recommended as the starting dose in pediatric patients with decreased renal function [see Clinical Pharmacology (12.3)]. Dosage Forms and Strengths Fexofenadine hydrochloride tablets USP are available in 30 mg, 60 mg, and 180 mg strengths. Fexofenadine hydrochloride tablets USP are coated with a peach colored film coating. Tablets have the following unique shape and identifiers: 30 mg tablets are capsule-shaped and have “93” on one side and “7251” on the other; 60 mg tablets are round and have “93” on one side and “7252” on the other; and 180 mg tablets are round and have “93” on one side and “7253” on the other. The changes in plasma levels were within the range of plasma levels achieved in adequate and well-controlled clinical trials. The mechanism of these interactions has been evaluated in in vitro, in situ, and in vivo animal models. These studies indicate that ketoconazole or erythromycin coadministration enhances Fexofenadine gastrointestinal absorption. This observed increase in the bioavailability of Fexofenadine may be due to transport-related effects, such as p-glycoprotein. In vivo animal studies also suggest that in addition to enhancing absorption, ketoconazole decreases Fexofenadine gastrointestinal secretion, while erythromycin may also decrease biliary excretion. Fruit Juices Fruit juices such as grapefruit, orange and apple may reduce the bioavailability and exposure of Fexofenadine. This is based on the results from 3 clinical studies using histamine induced skin wheals and flares coupled with population pharmacokinetic analysis. The size of wheal and flare were significantly larger when Fexofenadine hydrochloride was administered with either grapefruit or orange juices compared to water. Based on the literature reports, the same effects may be extrapolated to other fruit juices such as apple juice. The clinical significance of these observations is unknown. In addition, based on the population pharmacokinetics analysis of the combined data from grapefruit and orange juices studies with the data from a bioequivalence study, the bioavailability of Fexofenadine was reduced by 36%. Therefore, to maximize the effects of Fexofenadine, it is recommended that Fexofenadine hydrochloride tablets should be taken with water [see Clinical Pharmacology (12.3) and Dosage and Administration (2.1)]. Pregnancy Category C. There was no evidence of teratogenicity in rats or rabbits at oral doses of terfenadine up to 300 mg/kg (which led to Fexofenadine exposures that were approximately 4 and 30 times, respectively, the exposure at the maximum recommended human daily oral dose of 180 mg of Fexofenadine hydrochloride based on comparison of AUCs). In mice, no adverse effects and no teratogenic effects during gestation were observed with Fexofenadine hydrochloride at oral doses up to 3730 mg/kg (which led to Fexofenadine exposures that were approximately 15 times the exposure at the maximum recommended human daily oral dose of 180 mg of Fexofenadine hydrochloride based on comparison of AUCs). There are no adequate and well controlled studies in pregnant women. Fexofenadine hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Fexofenadine hydrochloride tablets are prescribed for the relief of symptoms of seasonal allergic rhinitis or for the relief of symptoms of chronic idiopathic urticaria (hives). Instruct patients to take Fexofenadine hydrochloride tablets only as prescribed. Do not exceed the recommended dose. If any untoward effects occur while taking Fexofenadine hydrochloride tablets, discontinue use and consult a doctor. Patients who are hypersensitive to any of the ingredients should not use these products. Patients who are pregnant or nursing should use these products only if the potential benefit justifies the potential risk to the fetus or nursing infant. Advise patients and parents/caregivers of pediatric patients to store the medication in a tightly closed container in a cool, dry place, away from small children. Advise patients and parents/caregivers not to take Fexofenadine hydrochloride tablets with fruit juices. Advise patients to take the Fexofenadine hydrochloride tablets with water.','url':'http://www.drugs.com/pro/fexofenadine.html','og_descr':'Fexofenadine official prescribing information for healthcare professionals. Includes: indications, dosage, adverse reactions, pharmacology and more.
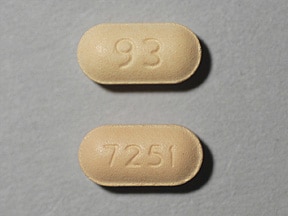
Allegra 60 mg tab. Oblong, peach, imprinted with E, 06. Fexofenadine 180 mg-MYL. Fexofenadine 60 mg-MYL. Round, blue, imprinted with M 753. Fexofenadine 60 mg.
ALLEGRA 60 mg tablets are available in: HDPE bottles of 100 NDC 0088-1107-47 with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner.


Nov 04, 2015 ALLEGRA fexofenadine hcl 60 mg tablets are available in: HDPE bottles of 100 NDC 0088-1107-47 with a polypropylene screw cap containing a.
 RSS Feed
RSS Feed
